Plastic recycling technology can be divided into two categories: physical methods (material recycling and thermal recycling) and chemical methods (chemical recycling). Among them, material recycling is relatively simple and low-cost. For example, the current recycling rate of PET bottles is more than 90%, but material recycling cannot avoid the degradation of the quality of repeatedly recycled plastic products, which greatly limits the recycling.
Chemical recycling can be divided into the gasification, oilification, and methanol recycling of waste plastics, and the monomer and oligomerization recycling of waste plastics. From a circular economy point of view, the latter has greater advantages. Chemical recycling is also feasible for the recycling of biodegradable plastics. This is because most biodegradable plastics are formed by the combination of ester bonds and amide bonds that are prone to hydrolysis, and are more prone to depolymerization, so monomer recovery is easier. Therefore, how to carry out biodegradable plastics Research on chemical recycling is also increasing.
Regardless of whether biodegradable plastics are microbially produced, natural or chemically synthesized, recycling should be pursued as much as possible. This is because biodegradable plastics are recycled, and the energy consumption and carbon dioxide production will be relatively small in the overall life cycle. In this regard, bio-based polymers such as polylactic acid (PLA) are the same. In the production of these substances, since the fermentation process and the chemical conversion process of the products require corresponding energy, it is more necessary to seek recycling-type recycling.
Recycling processes applicable to biodegradable plastics include reuse, material recycling, thermal recycling, chemical recycling, and biological recycling (Figure 8-1). At present, there are still many petroleum-based biodegradable plastics. In the future, with the development of biomass manufacturing technology, it is expected to gradually become bio-based biodegradable plastics. However, due to the limitation of resources, even for bio-based biodegradable plastics, continuous research is required to simplify manufacturing engineering and life cycle cycles, reduce the amount of moving materials, improve high-performance and high-skill materials, as well as recycling technology and corresponding system.

physical recycling
Material recycling is a method of maintaining the polymer state of plastics, melting and dissolving them, and then processing them into new products. Compared to plastics from petrochemical sources, biodegradable plastics should also be reused multiple times before material recycling. The difficulty in material recycling lies in the mixing of dissimilar polymers and additives, and the mixing of partially hydrolyzed polymers. Biodegradable plastics are the same as general-purpose plastics such as PET. After collection, they must be pre-treated such as impurity separation, selection, and pulverization, and then processed by melting and granulation. Generally, according to the different uses of plastics, the performance requirements will also be different. Therefore, due to the reasons mentioned above, the performance of plastics obtained from material recycling will be reduced, and they cannot be reused for the same purpose, but can only be downgraded.
Therefore, following the method of material recycling, it is necessary to consider chemical recycling. The popularity of biodegradable plastics is still very limited, so recycling is also limited. For example, the food utensils used in the 2005 Aichi World Expo in Japan, as an example of material recycling, were recycled and made into plant cultivation containers, which were used in Okayama Kokusai, Japan, which opened in October of the same year.
In actual use, in order to improve the properties such as strength and heat resistance, various additives and miscible solvents are mixed into PLA. For example, resins with sufficient strength and heat resistance can be obtained by mixing kenaf plant fibers into the casings of electrical appliances and auto parts. Kenaf itself grows very fast and has a large fixed amount of carbon dioxide, but it is rarely used as a plant fiber and is expected to be used as a bio-based fiber. The basic process of material recycling of PLA added with kenaf fiber is melting, granulation and re-molding, and there is no obvious physical property reduction during the process, so multiple material recycling can be carried out.
However, in the future, with the gradual increase in the variety and use of bioplastics, the amount of recycling that needs to be increased will also increase. It is obviously impossible to deal with material recycling alone, so chemical recycling and reuse are gradually being ignored by people.
chemical recycling
Chemical recycling is the process of chemically processing plastics into useful low-molecules for reuse. There are petrochemical-based energy recovery and monomer recycling and reuse of recycled plastic raw materials. There are many cases of utilization in the latter sense. From the perspective of reaction types, chemical recycling can be divided into two categories: thermal decomposition and chemical depolymerization. The methods of recycling plastics as chemical raw materials and fuels (thermal decomposition technology) are common thermal decomposition with isolated air and thermal decomposition under hydrogen. Attempts have been made to reduce the initial thermal decomposition to monomers in a vacuum, but during high-temperature thermal decomposition, side reactions are likely to occur in addition to the target main reaction, which is very difficult to control.
However, recently, by adding a catalyst to the thermal decomposition reaction, the reaction can be gradually controlled, and a precision depolymerization technology corresponding to the precision polymerization technology has been developed. The method of chemical depolymerization to generate monomers (depolymerization technology) can be divided into hydrolysis, alcohol decomposition, glucose decomposition, etc. according to different catalysts and solvents. In addition, biochemical processes using enzymes and microorganisms as renewable natural catalysts have also received attention.
The chemical structural sites at which the biodegradation and depolymerization of biodegradable plastics take place are basically common. Beginning with PLA and PHA, and later PCL, aliphatic polycarbonate, polyamic acid, etc., many biodegradable plastics have been found to have chemical recycling functions. Biodegradable plastics are mixed according to their physical properties, so separate chemical recycling by depolymerization is more effective than thermal decomposition. Moreover, as new technologies are developed to utilize ultra-(sub)critical carbon dioxide, water and methanol, the advantages of chemical recycling will also increase.
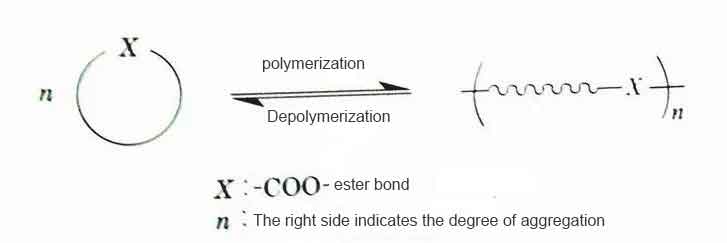
The basic reaction in the monomer reduction-type chemical recycling and reuse is a reaction that utilizes the equilibrium state of the polymerization reaction and the depolymerization reaction. In the ring-opening polymerization of cyclic monomers, the energy of ring-opening is the driving force for the polymerization. On the other hand, the polymerization consumes energy to make the equilibrium proceed in the direction of the cyclic monomers, thus forming a polymerization-depolymerization equilibrium state. That is, as shown in Figure 8-2, the ring-opening polymerization reaction is reversible with the change of reaction conditions, and the equilibrium is established. Therefore, in order to promote the depolymerization of the monomers, the produced monomers are vaporized by a method such as depressurization and isolated from the system. However, the thermal decomposition reaction generally carried out at high temperature is affected by side reactions and the structure of the terminal group of the polymer bond, so it is more difficult to control than precision depolymerization and precision polymerization.